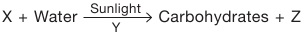Subject :NSO Class : Class 7
Subject :NSO Class : Class 8
Post Your Answer
Subject :NSO Class : Class 4
Post Your Answer
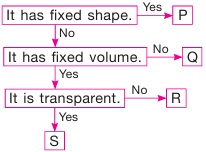
Subject :NSO Class : Class 6
Study the given flow chart and identify W, X, Y and Z.
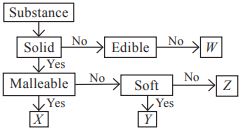
| W | X | Y | Z | |
| A | Honey | Graphite | Coin | Cooking oil |
| B | Dry ice | Plasticine | Sand | Iron nail |
| C | Mercury | Soap | Mirror | Cork |
| D | Petrol | Aluminium | Graphite | Diamond |
Post Your Answer
Subject :NSO Class : Class 4
Post Your Answer
Subject :NSO Class : Class 3
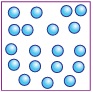
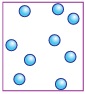
Observe the given pictures and select the option that correctly groups them.

| Pleasant sound | Unpleasant sound | |
|---|---|---|
| A | 1, 3, 5 | 2, 4, 6 |
| B | 2, 4, 5 | 1, 3, 6 |
| C | 1, 2, 3 | 4, 5, 6 |
| D | 4, 5, 6 | 1, 2, 3 |
what is happening in no.5
Post Your Answer
Subject :NSO Class : Class 3
Post Your Answer
Subject :NSO Class : Class 3
Post Your Answer
Subject :NSO Class : Class 6
Post Your Answer
Subject :NSO Class : Class 3