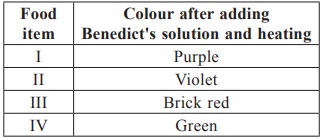
Subject :NSO Class : Class 6
Subject :NSO Class : Class 5
Refer to the given classification chart and select the option that correctly identifies P and Q.
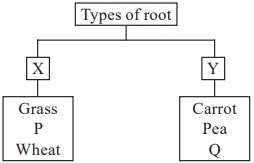
| P | Q | |
|---|---|---|
| A | Rice | Onion |
| B | Maize | Bajra |
| C | Onion | Beetroot |
| D | Bean | Radish |
Post Your Answer
Subject :NSO Class : Class 5
Ans 1:
Class : Class 6
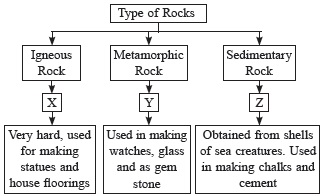
Granite is used for statues. Quartzite im not so sure but limestone is definetly obtained from the shells of sea creatures.
Post Your Answer
Subject :NSO Class : Class 6
Ans 1:
Class : Class 7
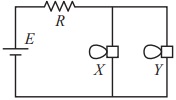
The answer should be D. When one bulb goes out in a parallel circuit, the brightness of the other bulbs remains unaffected, as each bulb has its own independent path for current flow, so the remaining bulbs continue to shine at their normal brightness; essentially, nothing changes to the brightness of the other bulbs.
Post Your Answer
Subject :NSO Class : Class 5
Post Your Answer
Subject :NSO Class : Class 7
As shown in the given circuit diagram, two identical heaters H1 and H2 are connected in series. Initially, switch S1 is closed and switch S2 is open and the reading of the ammeter and voltmeter are being recorded. Which one of following options correctly represents the changes in the readings of ammeter and voltmeter if switch S2 is closed after some time?
| Ammeter reading | Voltmeter reading | |
|---|---|---|
| A | Decreases | Decreases |
| B | Increases | Decreases |
| C | Increases | Increases |
| D | Decreases | Increases |
The circuit diagram is not given
Post Your Answer
Subject :NSO Class : Class 3
Post Your Answer
Subject :NSO Class : Class 6
Post Your Answer
Subject :NSO Class : Class 2
Post Your Answer
Subject :NSO Class : Class 3