Sanjana Iyer
from
HFS Powai, Mumbai
Subject :NSO Class : Class 6
Observe the given processes 1 and 2 carefully.
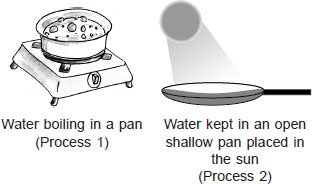
Which of the following statements are correct?
I. Bubbles are formed in process 1 while no bubble formation takes place in process 2.
II. Process 1 is slow while process 2 is fast.
III. Process 1 occurs at 100°C only while process 2 can occur at any temperature.
A I and II only
B II and III only
C I and III only
D All of these.
Being in a larger vessel, boiling of water in pan 1 should happen slower.
Ans 1:
Edit Post
Ans 2:
Edit Post
SOUVAGYA SAHA
from
De Nobili School, DHANBAD
Class : Class 9
YES !!!! SHOURYA GANESH IS A FOOLISH AND MAD FELLOW!!!!!
Ans 3:
Edit Post
Sumit Biswal
from
Dps gandhinagar, Gandhinagar
Class : Class 7
Only foolish Shourya not anyone else is foolish
Ans 4:
Edit Post
Sumit Biswal
from
Dps gandhinagar, Gandhinagar
Class : Class 7
Shourya is also a fool!! First he posted c as the answer which is actually correct and he is posting a which is wrong answer
Ans 5:
Edit Post
Sumit Biswal
from
Dps gandhinagar, Gandhinagar
Class : Class 7
You all are only fools except Shourya because boiling( Process 1) is fast and evaporation( Process 2) is slow!!! Please use some common sense and logic
Ans 6:
Edit Post
Shourya Ganesh
from
The Green Acres Academy, Mumbai
Class : Class 9
Answer should be (a). (Shourya Ganesh is a fool.)
Ans 7:
Edit Post
Aahan Jain
from
,
Class : Class 1
Shourya Ganesh posted that he is the fool only pls see the name on the top. but actually he is the fool


